Summary
Canadian women with breast cancer may choose breast conserving therapy as their course of treatment, requiring both breast conserving surgery and adjuvant radiation therapy. However, more than 15% of Canadian women fail to receive the appropriate radiation therapy, putting them at increased risk for recurrence. Age, distance from their radiation therapy centre and stage of disease affect patients’ likelihood of receiving prescribed radiation therapy. We propose a nomogram that allows physicians to predict which patients will and will not receive radiation. This nomogram, once validated, could be used to guide decision making when choosing between breast conserving therapy and mastectomy as the treatment course and thereby change the practice of breast cancer management.
Breast conserving therapy (BCT) is a mainstay of treatment for early stage breast cancer, consisting of breast conserving surgery (BCS) followed by adjuvant radiotherapy (RT). Breast conserving therapy carries equivalent survival for 20 years after surgery when compared with mastectomy; however, this success requires receipt of both the BCS and RT components of therapy.1 Receiving BCS alone is associated with an increased risk of tumour recurrence (5-fold increased risk of distant disease) and the need for additional treatments, such as reoperation and chemotherapy.2 This causes an increased financial burden on the health care system and patient. Yet, RT rates after BCS vary widely, with large U.S. studies reporting rates of approximately 85%.3 This phenomenon remains understudied in Canadian populations.
The decision between mastectomy and BCT for the management of early breast cancer carries important consequences for patient health and for the health care system. In an effort to understand the barriers to RT receipt, U.S. studies have identified factors associated with RT nonreceipt: increased age, medical comorbidity, increased distance from RT centre, high stage/grade, large tumour size, positive lymph node status, negative estrogen receptor status, in situ disease, and more. Insurance status has been identified as an important factor in receipt of RT in these studies; therefore, assessment of RT receipt in a universal health care system may help translate this knowledge into a Canadian context.4,5
We used data collected from 2006 to 2013 in the prospective breast cancer patient database in London, Ont., to create a cohort of all 1722 consecutive patients who received BCS for treatment of breast cancer. Variables included age at diagnosis, patient forward sortation area (FSA; the first 3 digits of the postal code), estrogen receptor (ER) status, progesterone receptor (PR) status, Her2Neu status, pathological stage, whether the tumour was invasive or in situ, and whether or not RT was delivered. Geographic access to treatment was calculated using the FSA and was defined as the time necessary to drive from the patient’s home to the nearest cancer centre offering RT, with cancer centres geocoded using ArcGIS 10.3. Time from each FSA to the nearest cancer centre was calculated using a road network based on route speed limits without traffic.
The cohort was divided into 2 groups: those who received RT, and those who did not. To determine the effect of case variables on whether or not a patient received RT, we performed logistic regression with backward elimination using R: a language and environment for statistical computing. We considered results to be significant at p < 0.05.
Of the 1722 patients who received BCS, 1455 received RT, while 267 did not. This produces a radiation receipt rate of 84.5%, meaning that 15.5% of patients fail to receive RT.
Time and age were significant covariates negatively associated with RT receipt (i.e., as age and time to RT centre increased, rate of RT receipt decreased). Disease stage had a variable association with RT receipt; compared with patients with in situ disease, those with early and metastatic disease (stage I, IIA, and IV) were less likely to receive RT and those with advanced non-metastatic disease (stage IIB and III) were more likely to receive RT.
We used the logistic regression data to produce a formula that calculates the likelihood (p) that a patient will receive RT following BCS:
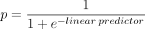
linear predictor = 7.6768 − (0.00961 × t) − (0.0251 × a) − (2.2854 × SI) − (1.8696 × SIIa) + (2.0277 × SIIb) + (2.2449 × SIII) − (4.4059 × SIV), where t = driving time from patient’s home to RT centre (min), a = patient’s age (yr), SI = 1 if stage I disease present (0 if not), SIIa = 1 if stage IIa disease present (0 if not), SIIb = 1 if stage IIb disease present (0 if not), SIII = 1 if stage III disease present (0 if not), and SIV = 1 if stage IV disease present (0 if not).
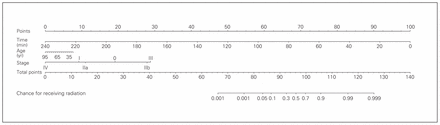
The magnitude of each covariate’s effect was used to produce a nomogram by rescaling variables to numbers between 0 and 100 (Fig. 1). Because the log (odds) of receiving radiation is additive regarding these covariates, their respective contributions are represented in the nomogram, and the sum is taken to determine a log (odds) and subsequently the odds of radiation receipt (Table 1). This nomogram predicts a given patient’s likelihood of receiving RT, which is valuable for clinicians, highlighting patients for whom extra encouragement, counselling and education on the importance of RT as an adjuvant therapy is warranted.
Nomogram for calculating the probability that a patient will receive adjuvant radiotherapy (RT).
Reference table for calculating the probability that a patient will receive RT*
This tool may make clinicians aware of the existing barriers faced by their patients, as clinicians and patients themselves may not be cognizant of the importance of these barriers at the initial consult. For instance, a clinician may not consider how specific distances, measured in minutes, from the cancer centre impact the chance of patients failing to receive complete treatment.
A more intriguing application of this tool is its use for patient selection. If a clinician determines preoperatively that a patient is unlikely to receive RT, the clinician might consult with radiation oncology before selecting BCT as a course of treatment or may recommend against BCT entirely, offering mastectomy instead. Such a tool makes the decision between BCT and mastectomy — a decision made by 1 in 9 women — evidence-based, rather than based on patient choice, especially when the patient has no pragmatic understanding of the treatment course. Finally, the tool may be used by investigators or granting agencies to guide patient selection for research, or by governments/payers to determine appropriate patients for BCT.
To our knowledge, this study is the first in Canada to highlight the unappreciated fact that 15% of patients undergoing BCS for breast cancer do not receive standard of care treatment, which includes radiation. This nomogram tool — the first of its kind — has the potential to change the practice of breast cancer management in Canada, and in so doing, improve the health of Canadian women and the health care system. The nomogram developed here should be externally validated in large administrative databases, at which point management recommendations could be developed for guidelines describing at what threshold to recommend BCT or advise against it, based on a patient’s likelihood of receiving RT.
Acknowledgements
The authors would like to thank Jingjia Chu and Dr. David Bellhouse from the Department of Statistics and Actuarial Sciences, Western University for their assistance with statistical analysis.
Footnotes
Competing interests: None declared.
Contributors: All authors contributed substantially to the conception, writing and revision of this article and approved the final version for publication.
- Received August 1, 2016.
- Accepted April 26, 2016.