Abstract
Background: Evidence regarding the safety and efficacy of intraoperative cell salvage (ICS) in transfusion reduction during cardiac surgery remains conflicting. We sought to evaluate the impact of routine ICS on outcomes following cardiac surgery.
Methods: We conducted a retrospective analysis of patients who underwent nonemergent, first-time cardiac surgery 18 months before and 18 months after the implementation of routine ICS. Perioperative transfusion rates, postoperative bleeding, clinical and hematological outcomes, and overall cost were examined. We used multivariable logistic regression modelling to determine the risk-adjusted effect of ICS on likelihood of perioperative transfusion.
Results: A total of 389 patients formed the final study population (186 undergoing ICS and 203 controls). Patients undergoing ICS had significantly lower perioperative transfusion rates of packed red blood cells (pRBCs; 33.9% v. 45.3% p = 0.021), coagulation products (16.7% v. 32.5% p < 0.001) and any blood product (38.2% v. 52.7%, p = 0.004). Patients receiving ICS had decreased mediastinal drainage at 12 h (mean 320 [range 230–550] mL v. mean 400 [range 260–690] mL, p = 0.011) and increased postoperative hemoglobin (mean 104.7 ± 13.2 g/L v. 95.0 ± 11.9 g/L, p < 0.001). Following adjustment for other baseline and intraoperative covariates, ICS emerged as an independent predictor of lower perioperative transfusion rates of pRBCs (odds ratio [OR] 0.52, 95% confidence interval [CI] 0.31–0.87), coagulation products (OR 0.41, 95% CI 0.24–0.71) and any blood product (OR 0.47, 95% CI 0.29–0.77). Additionally, ICS was associated with a cost benefit of $116 per patient.
Conclusion: Intraoperative cell salvage could represent a clinically cost-effective way of reducing transfusion rates in patients undergoing cardiac surgery. Further research on systematic ICS is required before recommending it for routine use.
Patients undergoing cardiac surgery often experience blood loss and coagulopathy requiring the administration of blood products to alleviate anemia, achieve hemodynamic stability and/or reduce the risk of ongoing bleeding. Despite this, blood transfusions have repeatedly been shown to be associated with increased morbidity and mortality.1–3 For this reason, strategies have been devised to reduce rates of transfusion in this population.
One such strategy is intraoperative cell salvage (ICS), the act of collecting shed blood throughout a surgical procedure, processing it via a cell saver device and reinfusing it into the patient during and/or at the end of the surgery. Direct transfusion of shed blood has been shown to be associated with a systemic inflammatory response,4 and the act of processing shed blood via filtration and centrifugation is felt to reduce the blood’s inflammatory potential5 and alter the need for transfusion. Despite these theoretical benefits, evidence as to whether ICS is safe and effective has been conflicting.6–19 Several studies have shown ICS to reduce exposure to allogeneic blood products with no changes in clinical outcomes or cost,14–16,19–22 while others have suggested that reinfusion with washed cell salvaged blood results in increased postoperative bleeding and heightened transfusion.17
The purpose of this study was to determine the effect of ICS on outcomes following cardiac surgery.
Methods
Study design
We retrospectively analyzed the cases of all patients who underwent nonemergent, first-time, on-pump cardiac surgery performed by a single surgeon 18 months before and 18 months after the implementation of ICS. Patients who refused blood products were excluded. The study was approved by the Horizon Health Research Ethics Board. Written informed consent to be included in the New Brunswick Health Centre registry was obtained from all patients.
Preoperative care
Antiplatelet agents other than acetylsalicylic acid (ASA) and anticoagulants, including warfarin, dabigatran, rivaroxaban and apixaban, were discontinued 5 days before the date of surgery. Conversely, ASA was continued up until the time of surgery.
Intraoperative procedure
All patients received general anesthesia using standard hospital practices. Patients were given a bolus of unfractionated heparin (400 units/kg) before cardiopulmonary bypass (CPB) and maintenance doses of unfractionated heparin as needed during CPB to maintain an activated clotting time (ACT) greater than 480 s. We measured ACT using Celite equivalent. The CPB circuit included a membrane oxygenator and centrifugal pumps to keep nonpulsatile systemic perfusion at flows of 2.4 L/min/m2. The CPB machine was primed with approximately 1660 mL of a solution containing 50 mL of 8.4% NaHCO3, 100 mL of 25% mannitol, 500 mL of 6% hydroxyethyl starch, 1000 mL of Plasmalyte, and 10 000 units of heparin. Tranexamic acid (Sandoz Canada Inc.) was infused at a rate of 1 mg/kg/h for 6–10 h following an initial bolus of 10–15 mg/kg bolus in all patients. Following separation from CPB, heparin was reversed with protamine (1 mg protamine/100 units of heparin). Modified ultrafiltration during CPB was not used.
Cell salvage procedure
We performed cell salvage in the ICS group using the Autolog autotransfusion system (Medtronic). The cell saver device processed blood collected from the surgical site to produce washed red blood cells (RBCs) for return to the patient during and/or at the end of the surgery. Cell salvage was used from the time of skin incision up until skin closure. Only when the patient was fully heparinized and on CPB were CPB suction catheters used preferentially where potentially large volumes of blood could be returned immediately to the CPB circuit. After the CPB, the patient received blood from the CPB circuit via the aortic cannula until hemodynamic stability was achieved. Cell salvage was resumed after administration of protamine. Once protamine had been administered and the aortic and venous cannulae were removed, all remaining pump contents were added to the cell saver reservoir and washed before being returned to the patient. At all times, cell saver blood was administered to the patient through a Lipiguard Filter (Terumo CVS), a 40 μm polyester screen-type lipid filter. A leukocyte-depleting filter was not used in this study.
By contrast, patients in the control group had shed mediastinal blood captured using a combination of CPB and wall suction catheters. Blood collected via CPB suction catheters was returned directly to the CPB circuit, while blood collected via wall suction catheters was discarded. At the end of the procedure, residual contents of the CPB circuit were directly reinfused into the patient following administration of protamine and upon transfer to the intensive care unit (ICU). Residual heparin in pump blood was reversed with additional protamine on admission to ICU.
Transfusion protocol
With respect to packed RBCs (pRBCs), an actual or anticipated hemoglobin level less than 70 g/L was treated with 1 or more units of pRBCs to maintain a hemoglobin level of 70 g/L or greater. No coagulation product transfusion protocol was used during the study. Coagulation products were administered in response to actual bleeding, perceived bleeding and/or a measured coagulopathy (e.g., elevated ACT, elevated international normalized ratio/partial thromboplastin time [INR/PTT] level or decreased platelet count). At no time were platelet function testing, fibrinogen assays and thromboelastography used to guide transfusion of coagulation products.
Data collection
Data were obtained from the New Brunswick Heart Centre Cardiac Surgery Database, a detailed observational clinical registry based on the core variables and data definitions contained within the Society of Thoracic Surgeons Adult Cardiac Surgery Database version 2.35. This registry prospectively collects pre-, intra- and postoperative data on all patients undergoing cardiac surgery at the Saint John Regional Hospital. Where lacking, additional data were collected through detailed chart review.
We collected data regarding the following baseline characteristics: age, sex, body mass index, smoking history, diabetes, dyslipidemia, renal failure (creatinine > 176 μmol/L), hypertension, chronic obstructive pulmonary disease (COPD), peripheral vascular disease, cerebrovascular disease, recent myocardial infarction (MI; ≤ 21 d), congestive heart failure, stable and unstable angina, atrial fibrillation, preoperative medications (ASA, angiotensin converting enzyme [ACE] inhibitors, β blockers and cholesterol-lowering agents), New York Heart Association (NYHA) class (IV v. I/II/III), ejection fraction (EF) lower than 40% and urgency status (elective v. urgent). In addition, we obtained data regarding the following intraoperative variables: type of surgical procedure, duration of CPB, aortic cross clamp time (AXC), administration of inotropes upon transfer to the ICU, and placement of intra-aortic balloon pump (IABP).
Primary outcomes
The primary outcome of interest was perioperative exposure to blood products. Perioperative blood product transfusion was defined as the administration of pRBCs and/or coagulation products, including fresh frozen plasma (FFP), platelets, cryoprecipitate, and factor eight inhibitor bypassing activity (FEIBA), either intraoperatively or within the first 24 hours after surgery. Both the percentage of patients receiving any of the aforementioned blood products and the volume of blood products administered per patient were considered. Volume of pRBCs, platelets and cryoprecipitate were measured in units. The approximate volumes per unit were as follows: pRBC 300 mL, platelet 200 mL and cryoprecipitate 10 mL. Fresh frozen plasma was measured in millilitres. We did not consider FEIBA volume owing to variability in dose per reconstituted vial.
Secondary outcomes
Secondary outcomes of interest included volume of chest tube drainage in the first 12 h postoperatively; rates of postoperative adverse outcomes (in-hospital mortality, reoperation for hemorrhage, infection, atrial fibrillation, renal failure, stroke, prolonged ventilation > 24 h and hospital length of stay [LOS]); pre- and postoperative hematological variables, including hemoglobin, INR/PTT and platelet count; and cost-effectiveness.
We estimated cost-effectiveness based on the reduction in volume of blood products administered among patients in the ICS group compared with controls after taking into account the expense associated with the use of the cell saver device. The cost, in Canadian dollars, of blood products was estimated as follows: pRBCs $425 per unit, FFP $716 per litre, platelets $603 per unit, cryoprecipitate $136 per unit, and FEIBA $1.46 per unit. Meanwhile, the cost of ICS was estimated at $202 per patient. The volume of blood products used per patient was multiplied by the cost per product. The total cost of blood products was then calculated for the ICS and control groups and then divided by the number of patients in each group to give an average cost of blood product use per patient. After factoring in the cost of the cell saver device among ICS patients, we then derived the average difference in cost between ICS and control patients to determine cost-effectiveness.
Statistical analysis
We made unadjusted comparisons between the ICS and control groups using the χ2 test or Fisher exact test for categorical variables. Continuous variables that followed a normal distribution were compared using t tests, whereas non-normally distributed continuous variables were compared using the Kruskal–Wallis test. We used multivariable logistic regression modelling techniques to adjust for other prognostically relevant baseline characteristics and intraoperative variables. Backward elimination was applied to 200 bootstrap subsamples to derive a parsimonious model for each outcome, including covariates retained in at least 50% of the subsamples. Bootstrapping also allowed for the estimation of the 95% confidence interval (CI) around the c-statistic from the 2.5th and 97.5th percentiles of the bootstrap distribution. We considered results to be significant at p < 0.05. All statistical analyses were performed using the SAS statistical software package, version 9.3.
Results
A total of 389 patients formed the final study population: 186 in the ICS group and 203 in the control group; 158 (40.6%) patients were 70 years of age or older, and 96 (24.7%) were women. Baseline characteristics were similar between the 2 groups, with the exception that those in the ICS group were more likely to present with unstable angina, NYHA class IV symptoms and an urgent status (Table 1). There were no differences in procedure type or the use of inotropes or IABP between the 2 groups; however, patients in the ICS group had shorter CPB and AXC (Table 2).
Baseline patient characteristics
Intraoperative variables
In the ICS group, the median amount of blood suctioned into the cell saver reservoir was 500 mL (interquartile range [IQR] 257.5–1000 mL), and the median amount of blood left over in the CPB circuit that was processed by the cell saver was 1000 mL (IQR 800–1200 mL), resulting in a median of 426 mL (IQR 317.75–600 mL) of concentrated RBCs to be transfused into the patient.
Primary outcomes
Concerning the primary outcome of interest, patients in the ICS group were less likely than controls to be exposed to pRBCs, coagulation products or any blood products in the perioperative period (Table 3). In addition, patients in the ICS group received a significantly lower volume of pRBCs (mean 0.75 ± 1.50 units v. 1.15 ± 1.79 units, p = 0.017) and FFP (mean 155 ± 433 mL v. 281 ± 450 mL, p < 0.001). The volume of platelets received by patients in the ICS group was also lower (mean 0.26 ± 0.66 units v. 0.36 ± 0.62 units, p = 0.10), but this difference did not reach statistical significance.
Perioperative exposure to blood products
Secondary outcomes
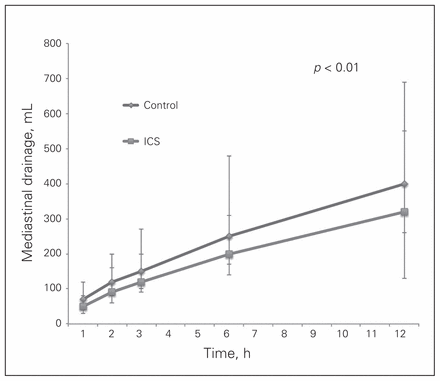
Regarding the secondary outcomes of interest, patients in the ICS group had less postoperative chest tube drainage in the first hour after surgery than those in the control group (50 v. 70 mL, p < 0.001), and this difference persisted over the first 12 h (median 320 mL [IQR 230–550 mL] v. median 400 mL [IQR 260–690 mL], p = 0.011; Fig. 1). No significant differences were noted in rates of postoperative adverse outcomes between the 2 groups (Table 4). Finally, despite having had similar hemoglobin, platelet and INR levels preoperatively, the ICS group had higher mean hemoglobin levels and lower median INR and platelet levels upon admission to the ICU than the control group (Table 5).
Chest tube drainage over time in the intraoperative cell salvage (ICS) versus the control group.
Postoperative adverse outcomes
Hematological variables
Following adjustment for baseline and intraoperative covariate risk factors, including prolonged duration of CPB, ICS emerged as an independent predictor of lower rates of perioperative RBC transfusion (odds ratio [OR] 0.52, 95% CI 0.31–0.87), coagulation product transfusion (OR 0.41, 95% CI 0.24–0.71) and transfusion with any blood product (OR 0.47, 95% CI 0.29–0.77). Full regression models are presented in Appendix 1, Table S1, available at canjsurg.ca. The median c-statistic for each of these 3 models derived using bootstrapping methods was 0.86 (95% CI 0.82–0.89) for pRBC transfusion, 0.79 (95% CI 0.73–0.84) for coagulation product transfusion and 0.84 (95% CI 0.80–0.88) for overall transfusion.
With respect to cost, the average cost of blood products per patient was significantly lower in the ICS group than in the control group ($604 ± $1194 v. $922 ± $1259, p = 0.011). After factoring in the cost of the cell saver device, an average savings of $116 was noted per patient when ICS was used.
Discussion
Intraoperative cell salvage was associated with decreased blood product transfusion, decreased postoperative bleeding, no differences in rates of postoperative adverse outcomes, and higher postoperative hemoglobin levels. Following adjustment for differences in baseline characteristics and intraoperative variables, ICS was independently associated with lower perioperative blood product transfusion rates. This decrease in blood product utilization translated into a significant cost savings per patient.
The decreased risk of perioperative pRBC transfusions found in patients who received ICS in our study compared favourably to findings from certain previously published studies8,12,14–16,19,22 but differed from others where either no change7,9,13,18 or an increase in perioperative pRBC transfusion rates17 was noted with the use of ICS. With respect to coagulation product transfusion, to our knowledge, the present study is the first to have shown a decrease in coagulation product transfusion with the use of ICS. This may reflect the relatively high transfusion rate of FFP (31.5%) and platelets (29.1%) in the control group, which may limit the applicability of these findings to other patient populations. Though this study did not demonstrate an overall reduction in morbidity associated with ICS, blood transfusions have been associated with poorer long-term outcomes, suggesting the reduction observed in this study is of clinical importance.1–3 Previous studies that have looked at ICS and the use of coagulation products have concluded that the removal of essential platelets and coagulation factors through cell salvage led to increased bleeding17 and greater coagulation product transfusion.9,17 While ICS was associated with lower postoperative platelet levels in this study, the absence of any deleterious effect of reduced postoperative platelet counts on chest tube drainage and blood product transfusion suggests that this hematological finding is of little clinical consequence.
The significant variation seen across studies in the effects of ICS on perioperative blood product transfusion and postoperative hematological parameters may reflect heterogeneity in how the cell saver was used in each study. In our study, the cell saver was used from the first incision until the end of the surgery, including the processing of residual contents from the CPB circuit after administration of protamine. Few ICS studies explicitly stated whether or not residual CPB contents were processed through the cell saver. Of those that did, it appears that the processing of residual CPB contents was associated with a significant decrease in pRBC transfusion12,14,15,19,22 and a decrease in postoperative chest tube drainage.19 The beneficial effect of ICS seen when processing residual CPB contents may result, in part, from the removal of excess heparin23 or an accumulation of inflammatory mediators commonly associated with CPB.24
We demonstrated an average cost savings of $116 per patient for whom ICS was used. This is in contrast to the results of Klein and colleagues,12 who found no cost advantage to using ICS. Varying methods of cost analysis as well as differences in the extent to which transfusion rates and volumes were reduced could have accounted for discrepancies in findings. Additionally, a minimum volume of blood processed has been shown to be necessary in order for ICS to be cost-effective.25 Studies in which residual CPB contents were not processed may not have observed the cost benefit that was appreciated in the present study owing to insufficient volume of blood processed. Regardless, the significant cost savings that we observed indicates that ICS is cost-effective.
Limitations
This study is not without its limitations. First, the retrospective nature of the study did not allow for complete elimination of confounding or bias in our analysis. Second, in the absence of strict coagulation product transfusion protocols at our institution, subjective differences in coagulation product transfusion practices over time may have played a role in the administration of blood products. By restricting our analysis to a single surgeon, we anticipate that some of this interpractitioner variability may have been eliminated. Third, as this study represents a pre- and postintervention analysis, it is possible that lower rates of transfusion following the institution of ICS was the result of increased experience and improved surgical technique on the part of the participating surgeon, as outcomes tend to improve as one gains experience.26 While we adjusted for the duration of CPB in the risk-adjusted analysis, adjusting for “evolution” in overall ability is statistically challenging and remains a limitation. Despite these limitations, the overall 18% reduction in blood products and, more specifically, the large 49% reduction in coagulation products are unlikely to be explained by change in surgeon experience or ability alone. Finally, the sample size of the study did not allow for risk stratification or examination of efficacy and cost-effectiveness of ICS in special high-risk groups. Undoubtedly, there may be increased cost savings in higher-risk surgical populations. However, the objective of this study was to examine the efficacy and cost-effectiveness of routine ICS in the general cardiac surgery population, and we were still able to demonstrate a benefit with ICS in this setting.
Conclusion
This study suggests ICS is a clinically and cost-effective method of reducing perioperative blood product transfusion in patients undergoing first-time cardiac surgery. These findings support the use of ICS as an agent of blood conservation in routine cardiac surgery. However, a prospective randomized multi-institutional controlled trial with evaluation of coagulation status and a strict transfusion algorithm is necessary to determine the true benefit of ICS.
Footnotes
Poster Presentation: Canadian Cardiovascular Congress, Vancouver, BC, Canada, Oct. 26, 2014
Funding: This work was supported by the Katelyn Robart Studentship through the Dalhousie University Summer Student Research Program 2013.
Competing interests: None declared.
Contributors: C. Côté, M. Ouzounian, C. Brown, R. Forgie and A. Hassan designed the study. C. Côté, J. MacLeod, B. O’Reilly and A. Hassan acquired the data, which C. Côté, A. Yip, J. Murray, M. Pelletier and A. Hassan analyzed. C. Côté and A. Hassan wrote the article, which all authors reviewed and approved for publication.
- Accepted July 4, 2016.