Abstract
Background: This study describes our experience with the placement of a skin-level gastrostomy device (MIC-KEY) in a single procedure.
Methods: We identified infants, children and young adults who underwent laparoscopic-assisted percutaneous endoscopic gastrostomy (LAPEG) tube insertion between October 2009 and June 2013. The steps of this procedure include upper endoscopy, single-port laparoscopy, gastropexy via percutaneous T-fasteners and placement of a skin-level gastrostomy device (MIC-KEY) using a “push” technique with a tear-away sheath.
Results: We included 92 patients in our study. Mean age was 3.7 years (range 3 wk–5 yr), and mean weight was 11.2 (range 2.8–54) kg. Median procedural time was 20 (range 12–76) minutes. Total median duration for the most recent 25 procedures was lower than that of the first 25 (62 v. 79 min, p = 0.004). There were no intraoperative complications or conversions to open surgery. Postoperative complications were observed in 6 (6.5%) patients. Three retained T-fasteners were assessed endoscopically (n = 1) or removed via local excision (n = 2). Two patients experienced early dislodged feeding tubes that were replaced via interventional radiology (n = 1) or repeat LAPEG (n = 1). There was also 1 intra-abdominal fluid collection that was drained percutaneously but ultimately required a laparotomy and washout. There were no major complications in the most recent 50 procedures.
Conclusion: Our results suggest that LAPEG is a safe, minimally invasive procedure for infants, children and young adults. This approach allows for immediate use of a skin-level gastrostomy device without the need for postoperative tube exchanges.
Gastrostomy tube insertion in children can be performed via open surgery, percutaneous endoscopic gastrostomy (PEG), laparoscopic gastrostomy, or percutaneous radiologic gastrostomy.1–5 Percutaneous endoscopic gastrostomy insertion remains popular in many centres because it can be performed quickly and does not require incisions.1,2 This approach provides visualization of the stomach but not the peritoneal cavity, which can lead to injuries to the colon, small bowel, liver and spleen.6–11 Another disadvantage is that the long PEG tube must be left in place for a few months before it can be exchanged for a skin-level device. This requires additional sedation, general anesthetic, or endoscopic retrieval. Another option is to remove the PEG tube in clinic, but this can be painful and unpleasant, especially for young children.
To avoid these issues, some surgeons have advocated using a laparoscopic approach, or a combination of laparoscopy and endoscopy.12–26 The combined approach is known as laparoscopic-assisted PEG insertion (LAPEG).5,12 This technique appears to be associated with reduced complications, but does not necessarily negate the need for postoperative tube exchanges for a skin-level device.5,12 As a result, there have been recent efforts to perform LAPEG in a single procedure.15 This approach requires a single anesthetic and allows for placement of a skin-level device that can be used immediately.
The purpose of the present study is to describe our experience with a 1-step LAPEG insertion using a skin-level device in, to our knowledge, the largest case series reported to date. We describe how this technique is performed and report our outcomes, including duration of surgery, length of stay in hospital (LOS) and complications.
Methods
Participants
This study received approval from the Health Sciences Research Ethics Board at Western University. All participants were treated by 1 of 3 pediatric surgeons at the Children’s Hospital, London Health Sciences Centre, between September 2009 and June 2013. Participants were identified prospectively and data were collected retrospectively from the electronic medical record.
Operative technique
Laparoscopic-assisted PEG insertion is performed in the operating room with the patient under general anesthesia. Necessary equipment includes a laparoscope, endoscope, MIC-KEY gastrostomy tube and MIC-KEY G introducer kit (Halyard Digestive Health).27 We used a 14-French MIC-KEY and introducer kit for infants and small children and a 16-French for older children and adolescents. The kit includes an introducer needle, guidewire, scalpel, dilator, tear-away sheath and 4 percutaneous T-fasteners (SAF-T-PEXY) with dissolvable monofilament suture material (Biosyn).
Following induction and intubation, the anticipated gastrostomy site is marked on the skin in the epigastric area. A medial location is chosen to prevent the gastrostomy site from migrating laterally as the child grows. The first assistant then inserts an appropriately sized endoscope into the oropharynx and advances into the stomach. The primary surgeon prepares the abdomen with a chlorhexidine solution and drapes in a sterile fashion. The unscrubbed first assistant operates the endoscope and remains positioned at the patient’s head.
The primary surgeon then makes a small incision below the umbilicus and inserts a 3 or 5 mm trochar and laparoscope using a Hasson technique. Once an adequate view is established, the laparoscope is operated by a second assistant. In practice, this person can be a medical student or the scrub nurse. The stomach is inspected laparoscopically, and a potential gastrostomy site is chosen along the greater curvature. Care must be taken to ensure that the gastrostomy is situated proximally enough to avoid obstruction of the pylorus and distally enough not to impede future Nissen fundoplication.
The primary surgeon then affixes the stomach to the abdominal wall using 2 or 3 percutaneous T-fasteners. The T-fasteners are passed first into the abdominal cavity (confirmed laparoscopically) and then into the lumen of the stomach (confirmed endoscopically). The use of laparoscopy prevents injury to intra-abdominal structures and ensures that part of the omentum does not become interposed between the stomach and abdominal wall. If this occurs, the gastrostomy tract may not mature properly and dehiscence may result. The use of upper endoscopy ensures that the T-fasteners are not passed “through and through” the anterior and posterior walls of the stomach.
The introducer needle is inserted between the T-fasteners and passed into the lumen of the stomach. A guidewire is inserted through the introducer needle, then the needle is removed, and a small incision is made on the skin at the guidewire exit site. The dilator and tear-away sheath are placed over the guidewire and passed into the stomach as a single unit using the Seldinger technique. The position of the tear-away sheath is confirmed endoscopically and the guidewire and plastic dilator are removed.
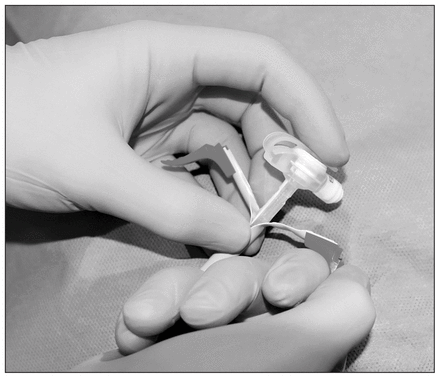
The balloon of the skin-level gastrostomy device (MIC-KEY) is tested with sterile water and deflated before placement. We use a MIC-KEY with a 1 cm stem for infants and small children and a 2 cm stem for older children and adolescents. Measurement of the tract length before insertion (to ensure a precise fit) is not required because the stomach is kept adherent to the abdominal wall by the T-fasteners rather than the gastrostomy balloon. The stem of the gastrostomy tube is lubricated with sterile jelly and then pushed into the gastrostomy site as the sheath is torn away (Fig. 1). After being advanced into the stomach, the balloon is inflated with sterile water. In general, only 3–5 mL of sterile water should be used, particularly with small children, where a large balloon can obstruct the pylorus.
Insertion of the skin-level gastrostomy tube (MIC-KEY) through the tear-away sheath.
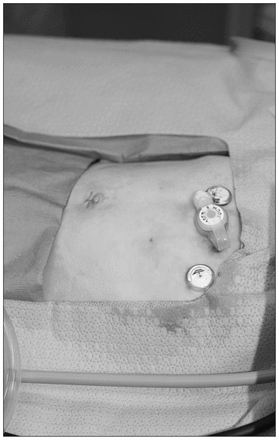
The position of the balloon is confirmed endoscopically, then the laparoscopic port and camera are removed, and the umbilical fascia is closed with a single dissolvable stitch. The skin is closed with a dissolvable stitch and covered with a dry dressing (Fig. 2). The gastrostomy site does not need to be closed with any additional sutures (other than then the percutaneous T-fasteners placed at the beginning of the procedure). We give all patients local anesthetic around the umbilicus at the end of the procedure as well as pre- and postoperative acetaminophen and ibuprofen. Patients may receive morphine postoperatively if required. Feeding can be started within 4 hours and advanced as tolerated.
Final result: small umbilical incision, secure T-fasteners and skin-level device (MIC-KEY) in place.
Statistical analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 21.
Results
Participant characteristics
We identified 92 infants, children and young adults who underwent LAPEG performed by 1 of 3 pediatric surgeons at our centre during our study period. Reasons for gastrostomy tube insertion included failure to thrive and/or oropharyngeal aspiration (90%), cystic fibrosis (8%), traumatic brain injury (2%) and oral aversion (1%). There were 45 male and 47 female patients, and their mean age was 3.7 ± 5.1 years (range 3 wk–25 yr). Their mean weight was 11.2 ± 8.8 (range 2.8 to 54) kg. Sixty-seven (73%) procedures were performed electively and 25 (27%) were performed on inpatients admitted to hospital for other reasons.
Outcomes
The median LOS for elective procedures was 4.3 ± 4.2 (range 1.4–31.3) days. The median skin-to-skin procedural time was 20 (range 12–72) minutes, the median anesthesia preparation and induction time was 18 (range 3–55) minutes, and the total median operating room time was 66 (range 30–126) minutes. Total operating room time decreased from a median of 79 minutes for the first 25 procedures to a median of 62 minutes for the most recent 25 procedures (p = 0.004).
Complications
There were no intraoperative complications or conversions to an open procedure. Six (6.5%) patients experienced postoperative complications that required repeat interventions. These included 2 (2.2%) patients who experienced early dislodged feeding tubes that required replacement. The first occurred approximately 3 weeks after insertion. There were no signs of leakage or peritonitis, so the patient underwent successful percutaneous radiologic gastrostomy under fluoroscopic guidance in the interventional radiology suite. The second patient had a gastrostomy tube that became dislodged 4 weeks after insertion. This child was brought to the emergency department, and a Foley catheter was placed in the gastrostomy site. A contrast study revealed a leak into the peritoneal cavity, so the patient was taken to the operating room and received repeat LAPEG with no further complications.
There was also 1 patient who experienced an intra-abdominal fluid collection and inflammatory response on postoperative day 2. The fluid was initially drained percutaneously but ultimately required a laparotomy and washout on day 7. The gastrostomy tube was in good position with no evidence of leakage and did not require a surgical revision. This patient was on acid suppression therapy before surgery and was not given preoperative antibiotics. As a result, the complex fluid collection was attributed to microscopic spillage of gastric contents during the initial LAPEG insertion. This was the only patient in this series (1.1%) who required a laparotomy.
Three (3.1%) patients had retained T-fastener material that was assessed endoscopically (n = 1) or removed via local excision (n = 2). The patient who underwent endoscopy presented with a deflated gastrostomy balloon approximately 1 month after LAPEG insertion. Upper endoscopy revealed an area of granulation tissue at the previous location of one of the T-fasteners but no visible suture material. This patient was ultimately switched to a nonballoon gastrostomy tube with a bolster and experienced no further complications. The 2 patients who underwent local excision for retained T-fastener material underwent surgery on an outpatient basis and did not require admission to hospital. These procedures occurred 8 months and 2 years, respectively, after initial LAPEG insertion.
Discussion
The first description of LAPEG in children was reported in 1995 in 2 adolescents in whom previous PEG insertion had failed.12 Many variations on this technique have been described since then.13–24 These include: the use of gastropexy versus no gastropexy, gastrostomy using a PEG tube versus a balloon-dependent device and “pull” versus “push” techniques to insert the gastrostomy tube. The LAPEG approach described here is similar to the Georgeson technique, which involves large, transabdominal U-stitches to affix the stomach to the abdominal wall.24 Gastrostomy tube insertion is then performed via sequential dilatation over a guidewire rather than insertion using a tear-away sheath. The main drawback of the Georgeson technique is that the U-stitches can slip, resulting in gastrostomy tract dehiscence and the need for reoperation.22,23
In our experience, LAPEG using a tear-away sheath is a safe and minimally invasive procedure for gastrostomy tube insertion in patients of all ages, ranging from infants to young adults. The rate of major complications appears to be lower than published rates for PEG tube insertion in children (5%–19%)2,9,10,21,22 and similar to those reported for the laparoscopic (2%–8%)9,10,20,21,23–26 and percutaneous radiologic techniques (4%–11%).4,28 Furthermore, LAPEG with a tear-away sheath allows for the placement of a skin-level device in a single procedure. As a result, additional visits to hospital for tube exchanges are not required. In children with complex conditions and multiple medical comorbidities, this represents an important benefit to patients and their families. The skin-level gastrostomy tube can be used immediately, and early feeding has not resulted in any complications. This results in a short hospital stay, with a median length of 4 days.
In addition, LAPEG with a tear-away sheath is associated with acceptable duration of surgery, which improves with increased experience. We found the dilator and tear-away sheath to be somewhat awkward at first, but the learning curve is short and handling improves after a few cases. Other drawbacks include the need for endoscopic and laparoscopic equipment in the operating room and 3 personnel to perform the procedure. Other techniques, such as laparoscopic gastrostomy, can be performed with a primary surgeon and single assistant.
Important safety benefits are gained from being able to simultaneously visualize the peritoneal cavity (via laparoscopy) and lumen of the stomach (via upper endoscopy). These include preventing injury to intra-abdominal structures, preventing the omentum from becoming interposed between the stomach and abdominal wall, accurate placement of the gastrostomy tube along the greater curve of the stomach, endoscopic confirmation of T-fastener placement and endoscopic confirmation of balloon inflation and position. This has resulted in no intraoperative complications or need for conversion to an open procedure.
All postoperative complications observed in our series occurred within the first 50 cases at our centre and within the first 40 cases for each surgeon. We have made small modifications to this technique, and there have been no further complications. The most important change was the decision to remove all T-fasteners on postoperative day 5 (we previously removed them in clinic 3 weeks to 1 month after surgery). Removal on day 5 prevents the suture material from becoming embedded in the subcutaneous tissue and causing chronic infection and granulation. The 2 patients with early dislodged tubes experienced these complications before making this change in practice, and we have not experienced any additional gastrostomy tract dehiscence following early T-fastener removal.
Conclusion
We believe LAPEG with a tear-away sheath is an excellent option for gastrostomy tube insertion. This approach is safe, fast and minimally invasive; provides adequate visualization; uses a skin-level device; allows for early feeding; and does not require tube exchanges postoperatively. This technique is associated with acceptable duration and rate of complications, and these outcomes appear to improve with increased experience.
Acknowledgements
The authors thank Brad Bell for his assistance with some of the photography included in this article.
Footnotes
The data presented in this study have been presented at the International Pediatric Endosurgery Group’s 23rd Annual Congress for Endosurgery in Children, July 22–26, 2014, Edinburgh, Scotland.
Funding: Michael Livingston is supported by the Clinician Investigator Program at McMaster University, funded through the Ontario Ministry of Health and Long-term Care.
Competing interests: None declared.
Contributors: M. Livingston, S. Jones, A. Bütter and N. Merritt designed the study. M. Livingston and D. Pepe acquired and analyzed the data. M. Livingston and D. Pepe wrote the article, which all authors reviewed and approved for publication.
- Accepted February 23, 2015.