Primary aortoenteric fistula (PAEF) is a relatively rare but serious complication of abdominal aortic aneurysm (AAA). Since Sir Astley Cooper first described PAEF in 1822, only about 300 cases have been reported in the literature. Roux and colleagues1 reviewed 253 cases before 1993, and Saers and Scheltinga2 analyzed 81 new cases from 1994 to 2003. As both studies report, PAEF is a clinical and surgical challenge associated with a high morbidity and mortality.
We report the case of a woman who presented with a PAEF and underwent a successful in situ reconstruction of the aorta. We describe the technical details that might decrease the chances of postoperative graft infection and fistula recurrence.
Case report
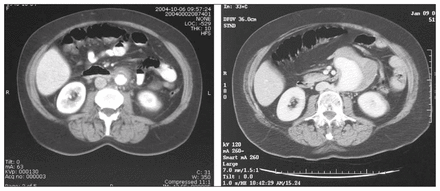
A 62-year-old woman presented with massive melena, hematemesis and brief loss of consciousness. Her surgical history included hysterectomy, bilateral oophorectomy, cholecystectomy and appendectomy. She had also undergone a sigmoid resection for recurrent episodes of diverticulitis a year before the current event; at that time, a preoperative abdominal computed tomography (CT) scan revealed a 3.3-cm infrarenal uncomplicated AAA (Fig. 1, left). After successful volemic resuscitation, a gastroduodenoscopy, performed as the initial diagnostic manoeuvre, revealed mild gastritis but no other anomaly. An abdominal CT scan revealed a thin-walled, saccular, inflammatory AAA, 7.5 cm in size, in intimate contact with the proximal jejunum (Fig. 1, right). Therefore we considered a diagnosis of PAEF and, in conjunction with our vascular surgery team, prepared the patient for surgery.
Preoperative computed tomography scans showing rapid enlargement of the abdominal aortic aneurysm, (left) 1 year before presentation and (right) at presentation.
Surgical exploration confirmed the radiologic findings. To ensure adequate vascular control and to minimize the risk of enteric contamination and post-operative graft infection, we took the following steps:
clamped the aorta, proximally and distally;
carried out proximal (at the level of the ligament of Treitz) and distal jejunal transection;
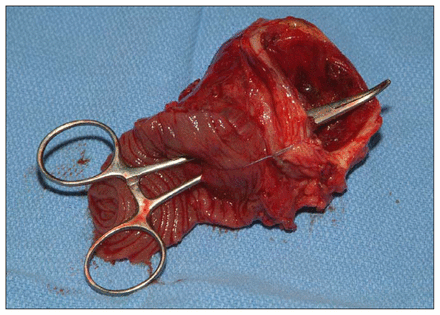
resected the aneurysm and bowel en bloc (Fig. 2);
performed aortoaortic reconstruction with a 16-mm Dacron graft;
carried out meticulous retroperitoneal closure;
performed digestive reconstruction with a transmesocolic side-to-side duodenojejunal anastomosis at the level of the second duodenum.
En-bloc resection of the aortic segment and small bowel.
The patient’s postoperative course was uneventful except for a transient biliary leak from a small tear in the liver capsule. We performed a water-soluble contrast study on postoperative day 6, which revealed a patent anastomosis; subsequently, the patient resumed oral feeding. The patient completed a 10-day course of antibiotics and was discharged on postoperative day 12. Results of serologic examination for Q fever were negative, as were blood culture specimens obtained preoperatively. Specimen cultures were also negative for tuberculosis and mycosis. Finally, histopathologic examination revealed acute inflammation around an atherosclerotic aortic aneurysm.
Discussion
The diagnosis of PAEF requires acute awareness. A minority of patients will present with the classical clinical triad of gastrointestinal bleeding, abdominal pain and a pulsating abdominal mass. Often, an initial, less significant, sentinel bleed is reported by patients before massive hemorrhage and exsanguination occur. Endoscopy is usually the first diagnostic tool to be used for upper gastrointestinal bleeding. However, as Saers and Scheltinga2 concluded, any negative endoscopy for gastrointestinal hemorrhage associated with AAA should be further evaluated by CT. In our case, the diagnosis was indeed entertained by the close proximity of the AAA and the bowel wall on CT, even though there was no obvious communication. In some cases, air bubbles in the aneurysm wall and intravenous contrast within the proximal gastrointestinal tract can facilitate the diagnosis.2,3 Most cases of PAEF are due to rupture of an atherosclerotic aneurysm into the bowel lumen. Traumatic, infectious (mycotic, tuberculous, syphilitic1), radiotherapy-associated and malignant4 causes have also been reported.
Despite the general consensus of the surgical community for early surgical treatment, most reports involve separation of the bowel from the aorta early in the procedure. We believe that en-bloc resection of the bowel and the AAA, when feasible, might lower the risk of gross contamination and graft infection. Furthermore, we suggest that localizing the enteric anastomosis as far as possible from the graft (in our patient, to the right side of the mesenteric root) further diminishes the risk of postoperative infection and recurrence of the aortoenteric fistula.
Although a few excellent long-term results have been reported in carefully selected patients who received treatment with in situ synthetic graft in such scenarios, clinical judgment is required. As most agree, an extra-anatomic vascular reconstruction should be favoured when facing diffuse retroperitoneal contamination. We performed aortoaortic synthetic grafting rather than an extra-anatomic bypass since there was no such contamination during the en-bloc resection. Moreover, the risk of aortic stump blowout after extra-anatomic reconstructions is significant. Finally, 5-year graft patency rates for aortoaortic bypasses (95%–100%) are much better than those reported for extra-anatomic bypasses (50%–80%). However, this must be weighed against the potential for graft infection, which occurs in 10%–20% of aortic grafts in similar situations compared with 3%–13% for axillobifemoral reconstructions.5 Endovascular treatment of such lesions might play a role in the future.2,6
Footnotes
Competing interests: None declared.
- Accepted April 21, 2008.