Abstract
The spleen is the most commonly injured visceral organ in blunt abdominal trauma in both adults and children. Nonoperative management is the current standard of practice for patients who are hemodynamically stable. However, simple observation alone has been reported to have a failure rate as high as 34%; the rate is even higher among patients with high-grade splenic injuries (American Association for the Surgery of Trauma [AAST] grade III–V). Over the past decade, angiography with transcatheter splenic artery embolization, an alternative nonoperative treatment for splenic injuries, has increased splenic salvage rates to as high as 97%. With the help of splenic artery embolization, success rates of more than 80% have also been described for high-grade splenic injuries. We discuss the role of computed tomography and transcatheter splenic artery embolization in the diagnosis and treatment of blunt splenic trauma. We review technical considerations, indications, efficacy and complication rates. We also propose an algorithm to guide the use of angiography and splenic embolization in patients with traumatic splenic injury.
The purpose of this paper is to review the current literature pertaining to transcatheter arterial embolization in the treatment of splenic injury. We discuss imaging, indications, technical considerations, clinical success and complication rates. We also propose an algorithm approach, including angiography and embolization, to managing patients with splenic injury.
The spleen is a bean-shaped organ located posterolaterally in the left upper quadrant of the abdominal cavity. The spleen is an important organ in the body’s immune system; it is the site where antibodies, monocytes and activated lymphocytes are produced. It constitutes a crucial defence against micro-organisms that enter the circulation.1 Because the spleen is highly vascular, splenic injury can be potentially life-threatening. This is particularly important because the spleen is the most commonly injured visceral organ in blunt abdominal trauma in both adults and children.2,3 Other less common causes of splenic injury include penetrating abdominal trauma, iatrogenic injury (e.g., surgery, endoscopy or biopsy) and spontaneous rupture.
Diagnosing and grading splenic injuries
The diagnosis of splenic injury following trauma is most frequently based on computed tomography (CT) scans. Numerous systems based on the extent of injury seen at CT, laparotomy or autopsy have been developed to grade traumatic splenic injuries.4–9 To standardize the reporting of splenic injuries, in 1994 the Organ Injury Scaling Committee of the American Association for the Surgery of Trauma (AAST) developed a grading system based on the anatomic disruption of the spleen, as shown on CT scans or during laparotomy (Box 1).4 The grading scheme is based on the estimated size of hematomas and lacerations, and it uses grades I–V to indicate the increasing severity of splenic injury. However, despite its widespread use, this system (along with similar, older CT grading systems) has been unreliable in predicting the outcome and guiding the management of blunt splenic injury.5,10–16 For example, the multi-institutional study of the Eastern Association for the Surgery of Trauma (EAST)17 reported failure rates of nonoperative treatment for each AAST grade of splenic injury; they reported a rate of 4.8% for grade I, 9.5% for grade II, 19.6% for grade III, 33.3% for grade IV and 75.0% for grade V. Recent literature has suggested that vascular injuries of the spleen (e.g., active splenic bleeding, pseudoaneurysms, post-traumatic arteriovenous fistulas) seen on contrast-enhanced CT scans are associated with an increased likelihood of failed nonoperative management. 9,18–21 However, these injuries are not included in the AAST grading system. One recent study by Marmery and colleagues22 compared the usefulness of the AAST splenic injury scale and a novel CT-based grading system that takes vascular injuries of the spleen into account. They found that the new system provided better discriminating ability when screening patients with blunt splenic trauma for arteriography or surgery than did the AAST injury scale. Thus, several new CT-based grading systems that take into account these major vascular findings have been proposed and adopted variably across some institutions.21,23,24
American Association for the Surgery of Trauma (AAST) guidelines for organ injury scaling for the spleen
| AAST grade* | Type | Description of injury |
|---|---|---|
| I | Hematoma | Subcapsular, < 10% of surface area |
| Laceration | Capsular tear, < 1 cm of parenchymal depth | |
| II | Hematoma | Subcapsular, 10%–50% of surface area Intraparenchymal hematoma, < 5 cm in diameter |
| Laceration | 1–3 cm in parenchymal depth not involving a parenchymal vessel | |
| III | Hematoma | Subcapsular, > 50% of surface area or expanding; ruptured subcapsular or parenchymal hematoma Intraparenchymal hematoma, > 5 cm in diameter |
| Laceration | > 3 cm parenchymal depth or involving trabecular vessels | |
| IV | Laceration | Laceration of segmental or hilar vessels producing major devascularization (> 25% of spleen) |
| V | Laceration | Completely shattered spleen Vascular hilar injury that devascularized the spleen |
↵* Advance 1 grade for multiple injuries to the same organ, up to grade III.
Management of traumatic splenic injuries
In the past 30 years, there have been significant advances in the management of traumatic splenic injuries. Although routine splenectomy for blunt splenic injury was once advocated, the risk of substantial postsplenectomy infection25–29 has motivated a shift toward spleen-preserving procedures and nonoperative management in patients who are hemodynamically stable.13,17,30,31 This change in the treatment of adult patients with splenic injury has been heavily influenced by experience in the pediatric trauma population.32 Currently, nonoperative management for blunt splenic trauma is the preferred treatment for the majority of patients who are hemodynamically stable, and it is the standard of practice in many centres.31,33
In general, standard nonoperative observational management commonly involves a period of bed rest, limited oral intake, serial hemoglobin and hematocrit measurements, and continued observation. Repeat CT scans are often performed 24–72 hours after initial injury; however, there is evidence that routine follow-up CT scans can be omitted in stable patients with blunt splenic trauma, AAST grade I–III, because they do not affect management.34–38 Instead, clinical symptomatology should indicate the need for additional imaging or intervention.38
Although nonoperative management has been shown to increase splenic salvage,39–41 failure necessitating further intervention may still occur. Failure rates for observational management have varied. In 2000, the multi-institutional EAST trial included 1488 patients with blunt splenic injuries, 61.5% of whom were managed nonoperatively. The failure rate for planned observation was 10.8%.17 However, a prospective study in 2003 reported an overall observational management failure rate of 34% and a rate of 44% for high-grade injuries (AAST grade III–V).42
In the past decade, angiography with transcatheter splenic artery embolization has played an increasing role in the nonoperative management of blunt splenic injuries. Numerous studies have demonstrated the method’s utility in increasing the success rate of nonoperative management, especially among patients with high-grade injuries.18,40,43–49 A recent study in 200641 compared 2 cohorts of patients (defined by 2 consecutive 7.5-year periods) with blunt splenic injuries that were managed nonoperatively. The authors found that the frequency of splenic artery embolization increased from 2.7% to 22.6% between cohorts, and the success of nonoperative management also increased from 77% to 96%, despite a higher mean injury severity score in the more recent cohort.41
Splenic arterial embolization
Transcatheter arterial embolization is performed by interventional radiologists. Sclafani50 first described its use in the treatment of splenic injuries in 1981. Since then, it has been adopted in many institutions to achieve increased rates of splenic salvage in patients with blunt splenic trauma.
Two primary techniques of splenic artery embolization have been described: proximal splenic artery embolization (PSAE) and superselective distal embolization. In PSAE, the splenic artery is selected with a catheter (with its tip at least beyond the origin of the dorsal pancreatic artery), and embolic coils are placed to occlude blood flow. The surgical equivalent of this procedure is splenic artery ligation, which was first reported in 1979.51,52 Proximal splenic artery embolization promotes hemostasis by causing a reduction in intrasplenic blood pressure, which may facilitate clot formation and the healing of the spleen.53,54 Splenic perfusion is likely maintained through a collateral arterial network, which develops rapidly after embolization.55–57
In distal embolization, a microcatheter is advanced as close as possible to the site(s) of vascular injury. Embolization is then performed using one or more small coils and/or pledgets of Gelfoam (Pfizer). This technique achieves hemostasis to the injured parts while preserving perfusion to the remainder of the spleen; however, subsequent bleeding may occur because some vascular injuries (e.g., pseudoaneurysms) may initially go unnoticed owing to vasospasm.46 This finding has been supported by data from Davis and colleagues,18 who reported that 74% of all the splenic artery pseudoaneurysms identified in their series were discovered on follow-up CT scans rather than on scans performed at the time of admission. Moreover, Smith and colleagues58 recently reported higher failure rates after distal embolization than proximal embolization (33% v. 22%). There is also some evidence suggesting that distal embolization may be associated with more frequent and larger splenic infarcts (as seen on CT scans) than proximal embolization.54,59
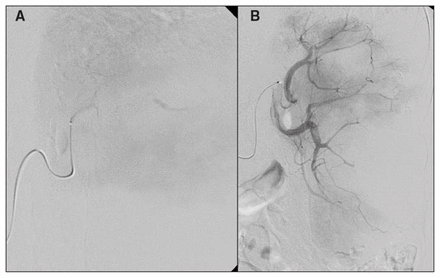
For these reasons, proximal embolization has recently been used more extensively than distal embolization for the management of blunt splenic injuries.44,47,54 Nevertheless, both techniques have been used successfully, sometimes combined in the same patient.47 The choice of technique and embolic agent is ultimately at the discretion of the radiologist performing the procedure (Fig. 1).
(A) Late-phase selective splenic angiogram through a microcatheter demonstrating contrast pooling and confirming active bleeding. (B) Selective splenic angiogram immediately postembolization demonstrating multiple perfusion defects. Contrast extravasation is no longer present.
Herewith, we present a review of the evidence supporting the use of transcatheter arterial embolization in select patients with blunt splenic trauma as a means by which to increase the proportion of patients who may be managed nonoperatively.
Indications for angiography and transcatheter arterial embolization
No universally accepted algorithm exists to determine the need for angiography and embolization. However, numerous authors reported on their management protocols for traumatic blunt splenic injuries. Early on, several authors — most notably Sclafani and colleagues,44,45 Hagiwara and colleagues48 and Haan and colleagues47 — proposed mandatory admission angiography for all hemodynamically stable patients with splenic injuries. The authors found that although contrast-enhanced conventional CT was accurate in diagnosing injury to the splenic parenchyma, it did not demonstrate splenic vascular injury (e.g., contrast extravasation, post-traumatic pseudoaneurysms and arteriovenous fistulas) well. All patients with proven vascular injury on angiography then underwent splenic embolization. However, in these earlier studies, the authors used now-obsolete CT and contrast enhancement techniques (e.g., small volumes of intravenous contrast, low infusion rates and slow scanning and scanning repetition times).20 In 1998, Davis and colleagues18 reported obtaining routine follow-up CT scans 48–72 hours after admission in all patients who were managed nonoperatively. Patients for whom CT scans showed evidence of vascular injury (on admission or at follow-up) underwent angiography, and all abnormal vessels were embolized.
Technologic improvements in the past decade have increased the accuracy of CT in identifying major splenic injuries. A study in 2006 found that CT had a sensitivity of 100%, specificity of 88% and overall accuracy of 93% in predicting the need for intervention.24 Authors of more recent studies advocate more selective use of angiography and embolization in the presence of the following CT findings: active contrast extravasation, splenic vascular injuries, AAST grade III–V injury and large hemoperitoneum.20,21,40,41,43,46,49,58,60
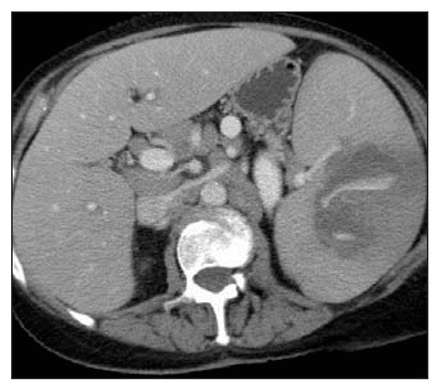
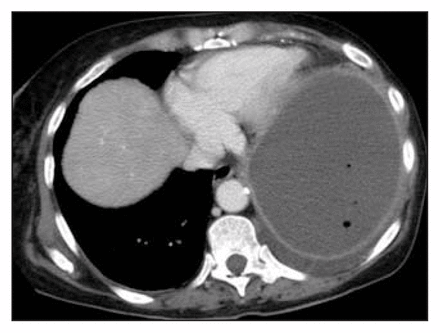
Active splenic hemorrhage is usually seen on contrast-enhanced CT scans as an irregular or linear area of contrast extravasation (Fig. 2). This may be seen in the splenic parenchyma, sub-capsular space or in the peritoneum.21 Post-traumatic splenic vascular injuries (e.g., pseudoaneurysms or arteriovenous fistulas) may be difficult to differentiate from active hemorrhage during the arterial phase, because both have similar attenuation values. However, unlike active hemorrhage, pseudoaneurysms and arteriovenous fistulas “wash-out” in the parenchymal phase, becoming isodense (or minimally hyperdense) relative to normal splenic parenchyma. Gavant and colleagues9 found that active splenic bleeding and traumatic splenic vascular injuries were both associated with a high failure rate (82%) of nonoperative management. Hence, several authors have successfully used splenic embolization in patients with active hemorrhage or vascular injuries to increase nonoperative splenic salvage rates.18,40,46,47,54,60 Similarly, splenic embolization has been used successfully in patients with AAST grade III–V splenic injuries, as discussed further in this paper (Table 1).40,43,54,60
Computed tomography scan, acquired during the venous phase of contrast injection, demonstrating an enlarged spleen with a low attenuation laceration containing high attenuation blood (active bleeding).
Efficacy of splenic embolization
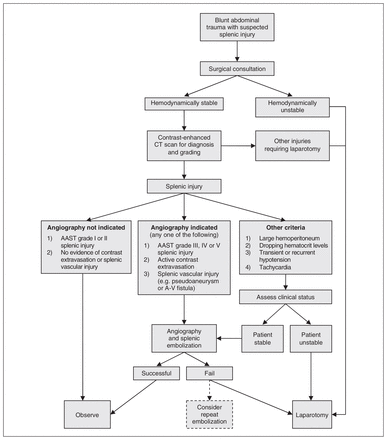
Herewith, we propose an algorithm to guide the use of angiography and embolization in blunt splenic trauma that attempts to combine the above findings with current state-of-the-art CT techniques (Fig. 3). All patients with blunt abdominal injuries and suspected splenic trauma initially require a surgical consultation as the first step in the management of their condition. Patients who are hemodynamically unstable should proceed directly to laparotomy. Those who are hemodynamically stable should undergo contrast-enhanced CT for the diagnosis and grading of splenic injuries. Patients with AAST grade I or II splenic injuries and no associated splenic vascular injuries can be managed with simple observation. Those who are found to have one of the previously mentioned CT findings indicative of angioembolization — including AAST grade III–V splenic injury, active contrast extravasation or vascular injury of the spleen — should proceed to angiography and splenic embolization. Consideration should be given to repeat embolization in patients who have failed initial embolization, because studies have shown this to be effective in increasing the overall success rate of nonoperative management. 46,47,49 Several institutions with well-coordinated surgical and angiographic services have successfully used splenic embolization in patients with less favourable clinical characteristics, including a large hemoperitoneum, recurrent hypotension despite responsiveness to fluid resuscitation, falling hematocrit levels with a progressive need for blood transfusions, and persistent tachycardia.43,49 Clearly, in such settings, good communication between the surgeon and the radiologist is the key to success. In at least one of these institutions, the attending surgeon supervises the angioembolization, the operating room is prepared at the same time and surgery can be performed immediately if splenic embolization fails or is abandoned in case of rapid clinical deterioration of the patient.43 Thus we offer an option in our algorithm for using splenic embolization in otherwise stable patients with one of these clinical characteristics. Naturally, success rates of splenic embolization in these settings would differ among institutions depending on the availability of angiographic services (including on-call), the level of integration of interventional and surgical services, and the degree of experience of the radiologists and surgeons.
Algorithm for blunt splenic injury. AAST = American Association for the Surgery of Trauma; A-V = arteriovenous; CT = computed tomography.
Efficacy of transcatheter arterial embolization
Since the first major study of the use of splenic embolization for blunt splenic trauma conducted by Sclafani and colleagues44 in 1995, several series have been published (Table 1). The success rate of splenic embolization ranges from 73% to 100%.18,40,43,44,46–49,54,58,60 The overall success rate of nonoperative management with the use of splenic embolization ranges from 86% to 100%,18,40,43,44,46–49,54,58,60 with most studies reporting success rates greater than 90%.18,40,44,47–49,54,60 Moreover, embolization has been shown to be useful in extending the type and number of splenic injuries that can be managed nonoperatively, as evidenced by the high success rates of nonoperative management (> 80%) for AAST grade III–V injuries.44,46,60 These results compare favourably with those of the EAST multicentre study,17 which did not involve splenic embolization and had an overall failure rate of 10.8% for nonoperative management, and even higher rates for high-grade injuries (19.6% for grade III, 33.3% for grade IV and 75.0% for grade V). Several authors also successfully re-embolized patients in whom initial embolization failed, which further increased the nonoperative splenic salvage rate.46,47,49
Safety of transcatheter arterial embolization
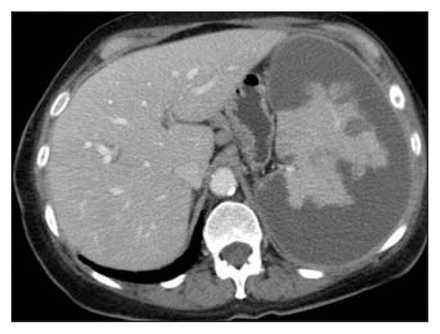
The Western Trauma Association multi-institutional trial46 was the largest study to date that examined complications following splenic artery embolization (n = 140). Major complications occurred in 19% of patients, and minor complications occurred in 23% of patients. There was some overlap between the 2 groups; a total of 32% of all patients had at least 1 complication. The most common major complication was blood loss, accounting for 11% of all complications; about half of these patients required a splenectomy because of persistent bleeding. Another common complication after embolization was splenic infarction (21% of all patients) (Fig. 4). This finding was previously well documented by Killeen and colleagues. 59 These authors noted that some infarcts contain gas (Fig. 5) and that, although this finding is of more concern because it makes it more difficult to exclude an abscess, most of these patients could be monitored clinically or with CT to ensure resolution. 59 However, the presence of an air/fluid level within a splenic sub-capsular fluid collection is a better predictor of an abscess. In such patients, percutaneous aspiration of the collection may be warranted to determine the presence of infection. Although most patients with splenic infarcts in the studies by Haan and colleagues46 and Killeen and colleagues59 were relatively asymptomatic and could be managed nonoperatively, the presence of an infarct could have explained the postembolization fever thereby easing concern for an additional source of infection. In the Western Trauma Association trial, 4 patients (3%) went on to develop splenic abscesses. Other relatively rare complications included coil migration, iatrogenic vascular injury and missed injuries to the diaphragm or pancreas. Similar findings have been described in other series.44,47
Computed tomography scan, acquired during the venous phase of contrast injection 15 days after embolization, showing residual enhancing splenic tissue within the surrounding infarcted spleen.
Computed tomography scan, acquired during the venous phase of contrast injection 15 days after embolization, showing a large fluid collection containing gas locules in the splenic bed.
Ekeh and colleagues61 examined complications following splenic artery embolization in 15 patients. They noted a major complication rate of 27%, which included splenic bleeding, splenic infarction, splenic abscess and contrast-induced renal insufficiency. Minor complications occurred in 53% of patients and included fever, pleural effusions and coil migration.
In 2005, Haan and colleagues60 reported only minor complications in the largest single-centre study of splenic embolization for blunt splenic trauma. These included splenic abscess, splenic infarct and coil migration.
In 2006, Smith and colleagues58 reported complications following splenic artery embolization in 41 patients. These included 3 procedural complications: a femoral artery dissection, a femoral artery arteriovenous fistula and a splenic artery dissection requiring splenectomy. Other complications included splenic abscess, deep vein thrombosis and symptomatic pleural effusion. Seven patients also experienced thrombocytosis but did not have adverse effects or require any treatment.
Splenic function postembolization
Following embolization, the primary long-term concern is the patient’s residual splenic immunological function and risk of infection. Asplenic individuals are most at risk of infection with encapsulated organisms. The lifetime risk of postsplenectomy sepsis is about 1%–2%.62 Most of these infections are caused by Streptococcus pneumoniae, Nesseria meningitides and Haemophilus influenzae. Thus it is generally recommended that patients who undergo splenectomy receive pneumococcal, meningococcal and H. influenzae (Hib) vaccinations. These should be given after the 14th postoperative day.62–69
Splenic function postembolization is an area of some debate because it has not been evaluated adequately in the literature. Theoretically, proximal splenic artery embolization may cause less impairment in splenic function than selective distal embolization because it allows the spleen to remain at least partially perfused, 55–57 thereby reducing the risk of infarction. A study of CT findings of embolized spleens found that proximal embolization was associated with less frequent and smaller splenic infarcts than distal embolization. 59 Also, several small studies demonstrated preservation of splenic function after surgical splenic artery ligation based on the absence of Howell–Jolly bodies and normal uptake of technetium-99 on scintigraphy. 52,70–73 Consequently, because proximal embolization mimics surgical splenic artery ligation, it is reasonable to theorize that it also does not impair splenic function.
Several small series with limited follow-up reported encouraging results regarding the preservation of splenic function after both proximal and distal embolization based primarily on scintigraphy.45,48 Bessoud and colleagues70 conducted the largest examination to date of splenic function after proximal embolization. They followed 24 patients for an average period of 26 months. They found Howell–Jolly bodies, indicating a functional impairment of splenic phagocytic function, in 2 of 24 patients. All patients assessed for exposure-driven immunity against H. influenza had sufficient immunity. Seventeen of the 18 patients explored for exposure-driven immunity against S. pneumoniae had sufficient immunity. Five of the 6 patients evaluated for pneumococcus vaccine response had a sufficient response. Although these findings are encouraging, large prospective studies are needed to more definitively evaluate splenic function after embolization and to rule out functional impairment. In the interim, it is our opinion that vaccination recommendations for embolized patients should be similar to those for patients undergoing splenectomy.
Conclusion
Transcatheter splenic artery embolization has a major role in the management of traumatic splenic injuries. Its addition to nonoperative management has clearly been shown to be feasible, safe and effective in increasing rates of splenic salvage. Embolization is particularly beneficial in injuries of AAST grade III or higher, which previously required laparotomy (with possible splenectomy) and were more likely to fail nonoperative treatment. Clinical and CT-based criteria can be used effectively to triage patients between simple observation, angioembolization and surgery. The success of splenic embolization is nevertheless based on good teamwork among surgeons and radiologists, as well as judicious selection of patients to undergo the procedure.
Footnotes
Competing interests: None declared.
Contributors: All authors contributed equally to the design of the study, acquisition and analysis of data, writing and review of the article, and each gave final approval for publication.
- Accepted March 11, 2008.