Abstract
Background: Minimal literature exists on outcomes for Canadian patients with gastroesophageal adenocarcinoma (GEA). The objective of our study was to establish a prospective clinical database to evaluate demographic characteristics, presentation and outcomes of patients with GEA.
Methods: Patients diagnosed with GEA were recruited from Jan. 30, 2017, to Aug. 30, 2020. Data collected included demographic characteristics, presentation, treatment and survival. A multivariable model for overall survival in patients treated with curative intent was created using sex, lymph node status, resection margin status, age and tumour location as variables.
Results: A total of 122 patients with adenocarcinoma of the stomach or gastroesophageal junction were included. Median age was 65 years (interquartile range [IQR] 59–74), 70% of patients were male and 26% were born outside of Canada. Median follow-up time was 14.5 (IQR 8.0–31.0) months. Following staging computed tomography scanning, 88% of patients were deemed to have potentially resectable disease. Eighty-one (76%) received staging laparoscopy and 74 (61%) were treated with curativeintent surgery. Forty-six (62%) patients had nodal metastases. The median number of nodes harvested was 22 (IQR 18–30). The R0 resection margin rate was 82%. The 3-year overall survival for patients who received curative-intent treatment was 63% and 38% for all patients. On multivariable analysis, female sex (hazard ratio [HR] 3.88, p = 0.01), positive nodal status (HR 3.58, p = 0.02), positive margins (HR 3.11, p = 0.03) and tumour location (HR 3.00, p = 0.03) were associated with decreased overall survival.
Conclusion: Many of the patients with GEA in this study presented with advanced disease, and only 61% were offered curative-intent surgery. A prospective multicentre national GEA database is now being established.
Gastroesophageal adenocarcinoma (GEA) is a common cancer worldwide and a major cause of cancer-related deaths.1 Previous literature clearly demonstrates differences in the presentations and disease pathology seen across Asian and North American populations, with North American patients often presenting with later stage disease, leading to worse overall outcomes.2–4 Most of the literature on the epidemiology of GEA in North America is from the United States, and there is a relative paucity of Canadian data. Available literature indicates that GEA leads to the death of approximately 1950 Canadians annually.5
To assess outcomes for patients in our region with GEA, we developed a prospective clinical database to better understand this disease in a Canadian context. The aim of our study was to evaluate how patients with GEA in Alberta present, the types of treatment they receive and their outcomes. Findings from this study will be used to improve the pathway of care at our institution. This work has also led to the development of a national prospective gastroesophageal cancer database involving multiple academic centres across Canada, which will be used to improve care for Canadian patients.
Methods
Database creation
Variables of interest were determined by a focus group that included surgeons and gastroenterologists. A patient consent form was developed with approval from the Alberta Research Ethics Community Consensus Initiative (ARECCI) and the Health Research Ethics Board of Alberta Cancer Committee.
Patient recruitment started on Jan. 30, 2017, and ended on Aug. 30, 2020. The secure Web-based software REDCap (Vanderbilt University) was used to collate patient information.6,7 At the initiation of our study, an invitation was sent to all surgeons who treat GEA in Alberta, Canada, to participate in the recruitment of patients. The end date was when the national prospective database on gastroesophageal cancer was established.
Data collection
This is a prospective cohort study of patients diagnosed with GEA in Alberta. We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for the reporting of observational studies.8 All patients diagnosed with GEA in Alberta were eligible for inclusion. Patients were referred to the study by participating health care providers. The majority of the patients were from northern Alberta, and patient recruitment occurred across 4 hospitals. Data were obtained by providers from patients and entered manually into the REDCap database. Follow-up was completed by reviewing electronic medical records and contacting patients by phone. The last date of follow-up in regard to survival was Oct. 11, 2021.
Demographic data collected included age, sex, race, country of birth, presence of a family history of gastroesophageal cancer, body mass index at time of diagnosis and smoking history. Data on medical comorbidities were recorded and all patients were assigned a Charlson Comorbidity Index score.9,10
Variables were separated into 3 primary categories: presentation, diagnosis, and treatment and outcomes. Presentation variables of interest included initial symptoms, wait times to investigation and location of tumour. Diagnostic variables of interest focused on staging investigations including resectability status on initial computed tomography (CT) scan and results of staging laparoscopy. Treatment outcome variables included details on neoadjuvant, surgical and adjuvant therapy as well as postoperative outcomes such as length of hospital stay, rate of postsurgical complications, final pathology details and survival.
On the basis of the results of the staging CT scan of the chest, abdomen and pelvis, the treating surgeon was asked to classify each tumour as resectable, borderline resectable or unresectable for curative intent. Patients whose CT scan results indicated obvious vascular involvement, ascites, omental nodules, peritoneal nodules, liver metastases or distant lymph node metastases were judged to have unresectable tumours. Examples of borderline resectable cases included those with very large tumours, multiple enlarged but potentially resectable lymph nodes, and close proximity of tumours to major vascular structures without clear involvement. In 2 patients with limited peritoneal disease, cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (HIPEC) was performed to remove all macroscopic disease and treat the remainder of the peritoneal cavity with chemotherapy. Details for these patients are reported separately.
Data analysis
Data were analyzed by a biomedical statistician (S.G.) using SPSS version 25 (IBM). A descriptive analysis was completed for the patients’ demographic and clinical data. Separate analyses were completed for the entire cohort (122 patients) and for the subgroup of patients treated with curative intent (74 patients). Categorical data were expressed as frequencies and proportions while continuous data were expressed as medians with interquartile ranges (IQRs). Time to event analysis for overall survival (OS) and recurrence-free survival (RFS) was conducted using the Kaplan–Meier method.11 Three-year survival probabilities for OS and RFS were reported. The log-rank (Mantel–Cox) test was used to compare the survival curves. To identify the factors associated with overall survival, Cox hazard regression analysis was conducted. Hazard ratios (HRs) and the corresponding 95% confidence intervals were reported.12 A mutivariable Cox model for OS among patients treated with curative intent included sex, lymph node status, resection margin status, tumour location (gastroesophageal [GE] junction v. other) and age. A binary logistic regression model was used to identify the factors associated with positive staging laparoscopy (yes v. no). The preoperative variables included in the multivariable logistic regression model included age, sex, weight loss status, tumour location, visibility of the tumour on staging CT (yes or no) and the presence of enlarged lymph nodes on staging CT. Odds ratios (ORs) and 95% confidence intervals (CIs) were reported. Statistical significance was set at a p value of less than 0.05.
Measures to minimize bias
To minimize bias in our study, we did not exclude any specific patient groups. Medical professionals obtained medical information directly from their patients, in a timely fashion. Data were collected prospectively, and patients were recruited near the time of diagnosis and before treatment, minimizing the risk of recall bias.
Results
Presentation
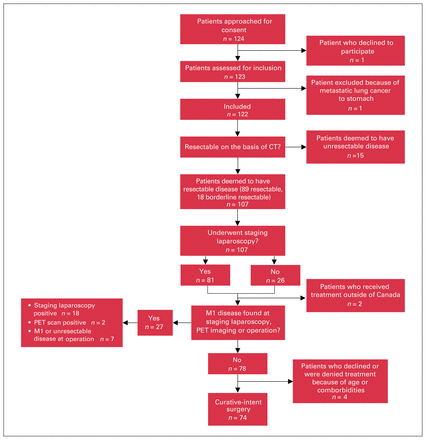
A total of 124 patients were approached, of whom 123 agreed to participate. One patient with stage 4 disease at presentation declined to participate (Figure 1). One patient who presented with a perforated gastric malignancy was excluded after pathology revealed metastatic lung cancer, leaving 122 cases for analysis.
Study flow chart. CT = computed tomography; PET = positron emission tomography.
Characteristics of the included patients are presented in Table 1. The median age at the time of diagnosis was 65 (IQR 59–74) years, and 70% of the patients (n = 85) were male. With regard to race, 93 (76%) patients were White, 18 (15%) were Asian, 8 (7 %) were First Nations or Métis, 2 (2%) were African and 1 (1%) was South or Central American. Only 12 (10 %) patients reported a first-degree family history of GEA. The majority (63%) of the patients had a smoking history of at least a 1 pack-year, with 48 (39%) reporting a history of more than 20 pack-years.
Demographic and clinical characteristics of patients at presentation
Despite 45 (37%) patients having lost 9 kg or more at presentation, 67 (55%) were classified as overweight or obese at diagnosis. Weight loss was more pronounced among the patients who were unable to receive curative-intent surgery, with a mean weight loss of 9.1 kg (standard deviation [SD] 9.5 kg) compared with 5.4 kg (SD 8.6 kg) in the patients treated with curative intent.
The most common presenting symptom in our cohort was dysphagia or odynophagia, in 38 (31%) of the 122 patients, followed by abdominal pain (21 patients, 17%), new-onset gastroesophageal reflux disease (19 patients, 16%) and anemia (16 patients, 13%). Less common presenting symptoms included melena (10 patients, 8%), weight loss (7 patients, 6%) and hematemesis (3 patients, 2%). Six (5%) patients had tumours that were found incidentally while doing investigations for other pathologies (Table 1). The median number of days of related symptoms before diagnosis was 128 (IQR 65–247) days.
Diagnosis
Of the 122 tumours, 53 (43%) were classified as proximal, involving the GE junction or gastric cardia. Sixty-one patients (50%) had distal tumours involving the gastric body, antrum or pylorus, and 8 patients (7%) had diffuse malignancy involving the entire stomach. Only 91 (75%) of the biopsy reports commented on the Helicobacter pylori status; 12 (13%) patients were found to have positive results. Forty-six (38%) patients had signet ring cells present on initial biopsy (Table 1).
On staging CT scan, 101 (83%) patients had visible tumours, 62 (51%) had enlarged lymph nodes and 13 (11%) had evidence of metastatic disease, with 2 (2%) of these patients having metastatic disease affecting the lungs. On the basis of initial CT scanning, 89 (73%) patients were judged to have resectable tumours, 18 (15%) patients had borderline resectable tumours and 15 (12%) had unresectable tumours (Table 1).
Of the 107 (88%) patients who had potentially resectable tumours, 81 (76%) received staging laparoscopy. Intraperitoneal samples were taken for cytology in 75 (93%) patients. Nine (11%) patients were found to have peritoneal disease and positive (n = 7) or suspicious (n = 2) cytology, 8 (10%) patients had obvious metastatic disease and either negative or incomplete cytology results, 4 (5%) patients were found to have no obvious peritoneal disease but suspicious or indeterminate cytology results and 1 (1%) was found to have positive cytology results and no obvious peritoneal disease (Table 1). Of the 4 patients with no obvious metastatic disease but suspicious cytology results, 3 had their tumours resected and all 3 experienced recurrent disease, surviving 463, 726 and 1083 days after their operation, respectively. One patient with suspicious cytology results as well as the 1 patient with positive cytology results but no obvious peritoneal disease were found to have unresectable metastatic cancer on positron emission tomography (PET) scanning and did not receive a curative operation. Multivariable analysis of preoperative variables found that tumour involvement of the entire stomach was the only variable significantly associated with finding M1 disease at staging laparoscopy (Table 2).
Multivariable regression for positive results on staging laparoscopy for 81 patients
Treatment and outcomes
Of the 107 patients deemed to have potentially resectable tumours after staging CT, 50 (47%) were discussed at multidisciplinary rounds and 42 (39%) received pre-operative chemotherapy. There were a variety of reasons why patients did not receive neoadjuvant chemotherapy: 20 (19%) patients were found to require palliative care; 20 (19%) patients were not candidates because of comorbidities; 11 (10%) patients required urgent surgery for obstruction, bleeding or gastric perforation; 8 (7%) patients refused neoadjuvant therapy; and 6 (6%) patients had early disease not requiring chemotherapy.
Of the 42 patients who received neoadjuvant treatment, 30 (71%) patients received 5-fluorouracil, leucovorin, oxaliplatin and docetaxel (FLOT), 7 (17%) received carboplatin and paclitaxel as per the protocol from the Chemoradio-therapy for Oesophageal Cancer Followed by Surgery Study (CROSS), 4 (10%) received epirubicin, cisplatin and 5-fluorouracil (ECF) and 1 (2%) received folinic acid, fluorouracil and irinotecan (FOLFIRI).13–16 Thirty-eight of the 42 patients (90%) completed their neoadjuvant treatment. Seven patients (17%) received preoperative radiotherapy, all of whom had GE junction tumours.
With regard to surgical procedures, 84 (69%) patients were taken to the operating room for a procedure other than staging laparoscopy. Only 74 (61%) patients received curative-intent surgery in Edmonton, 2 patients received hyperthermic intraperitoneal chemotherapy (HIPEC) and 1 patient returned to their home country to receive curative-intent surgery. Seven patients were found to have unresectable disease at the time of operation, with 2 of these patients receiving a palliative bypass and 1 receiving a palliative resection. Two of these 7 patients (29%) found to have unresectable tumours had not received staging laparoscopy. Of the 84 procedures, 80 (95%) were completed by surgeons with surgical oncology or thoracic surgery fellowships. The patients in the study were treated by 11 surgeons.
The most common resection performed was a subtotal distal gastrectomy (35 patients, 47%), followed by total gastrectomy by an abdominal approach (23 patients, 31%). Less common procedures included total gastrectomy by a thoracoabdominal approach (5 patients, 7%), esophagectomy with a right chest anastomosis (7 patients, 9%), esophagectomy with a neck anastomosis (2 patients, 3%) and total esophagogastrectomy with a colonic interposition (2 patients, 3%). All procedures were performed using an open surgical approach. R0 margins were achieved in 61 (82%) patients. The median number of lymph nodes harvested was 22 (IQR 18–30) and 80% of patients had more than 15 lymph nodes sampled (Table 3).
Treatment characteristics of patients who received curative-intent treatment
Postoperatively, the median length of stay was 10 (IQR 8–16) days. When we assessed complications using the Clavien–Dindo scale, 7% of patients had grade 4 complications, 11% had grade 3 complications and 19% had grade 2 complications.17 One patient (1%) died within 30 days of surgery following a myocardial infarction, and 90-day postoperative mortality was 3% (Table 3).
Hyperthermic intraperitoneal chemotherapy
Two patients had limited peritoneal disease at staging laparoscopy and received FLOT neoadjuvant chemotherapy, gastric resection, cytoreduction therapy and HIPEC. The postoperative lengths of stay for these patients were 30 and 11 days, respectively. The first patient experienced a recurrence 11 months after surgery and died 27 months after surgery. At the time of writing, the second patient was alive with no evidence of disease 2 years after surgery.
Overall and disease-free survival
No patients were lost to follow-up. The 3-year OS for all patients in the study (n = 122) was 38%. Median survival was 28 (95% CI 14–42) months. For the 74 patients who underwent curative-intent surgery, 3-year OS was 63%, the median survival time was 46 (95% CI 35–57) months and the 3-year RFS was 60%.
Multivariable Cox proportional hazards model for overall survival
On multivariable analysis, decreased OS in patients treated with curative intent was significantly associated with female sex (HR 3.88, 95% CI 1.43–10.58, p = 0.01), the presence of nodal disease (HR 3.58, 95% CI 1.18–10.85, p = 0.02), a positive resection margin (HR 3.11, 95% CI 1.15–8.40, p = 0.03) and tumour location (GE junction, HR 3.00, CI 1.11–8.12, p = 0.03; Table 4). In our multivariable model, age at diagnosis was not significantly related with decreased OS (HR 1.02, 95% CI 0.98–1.06, p = 0.30). Median follow-up time was 14.5 (IQR 8.0–31.0) months. The multivariable analysis including all 122 patients is shown in Appendix 1, Supplemental Table 1, available at www.canjsurg.ca/lookup/doi/10.1503/cjs.0051221/tab-related-content.
Multivariable Cox proportional hazards model for overall survival in 74 patients who underwent curative-intent treatment
Discussion
To the best of our knowledge this study represents the first prospective Canadian database for patients with GEA. Our results are similar to those of studies performed at other centres in North America, with many patients presenting with late-stage disease.18 The reason for late-stage presentations is probably multifactorial, but the median duration of symptoms before diagnosis was approximately 4 months, which may be due to physician inexperience with GEA, which is relatively rare in Canada. There is also considerable overlap between the symptoms of GEA and those of very common benign conditions (e.g., gastroesophageal reflux disease), which makes the job of primary care physicians very challenging. Only 61% of our patients were able to undergo curative-intent resection; for these patients, 3-year OS was 63%, 30-day postoperative mortality was 1% and 90-day postoperative mortality was 3%, suggesting that for patients diagnosed with nonmetastatic GEA, effective multidisciplinary treatment with long-term survival is possible.
We found numerous modifiable risk factors for GEA in our patient population, including smoking, obesity and H. pylori infection, suggesting a role for primary care interventions to reduce the incidence of this disease in Canada.19–22 Looking at high-risk populations in our study, 26% of our patients were born outside of Canada, supporting previous research suggesting that first-generation immigrants from at-risk countries remain at high risk for GEA.23 Previous research by Kim and colleagues has even suggested that “endoscopic screening for gastric cancer in Korean Americans should be considered” because of their significantly increased risk compared with non-Hispanic White patients.24 This practice is not routinely followed in Alberta, and as far as we are aware it is not practised elsewhere in the country.
Our study included a high proportion of patients with proximal tumours, with 43% being found at the GE junction or gastric cardia, similar to the findings of previous North American studies.2,25,26 In Alberta, 29% of adults are classified as overweight or obese; this may be correlated to the high rate of proximal tumours in our study, as obesity has been previously reported to be linked to the rising incidence of GE junction adenocarcinoma.27–30 Also, in the initial biopsy signet ring cells were found in more than one-third of patients, which is comparable to the findings of previous Western studies and higher than the figure quoted in many Eastern studies.31–33 Signet ring cell carcinoma is associated with poor prognosis in patients with GEA, and this may be linked to our finding that it was possible to offer curative-intent treatment to only a relatively low proportion of patients.34
Given the high incidence of advanced disease at presentation in North American patients, it is critical for surgeons to conduct appropriate clinical staging pre-operatively to inform decision-making on the best treatment for patients. Although CT of the chest is recommended in most staging guidelines for GEA, the true incidence of metastatic pulmonary disease at diagnosis is quite low.35,36 In our cohort, only 2% of patients had clear lung metastases at their staging CT scan. In a retrospective study, our group previously found an incidence of pulmonary metastases of 5.4%.37
In a landmark study from Memorial Sloan Kettering Cancer Center in 1997, Burke and colleagues demonstrated that staging laparoscopy spared 24 of 111 patients with gastric cancer the morbidity associated with undergoing a full laparotomy, as these patients were found to have M1 disease at laparoscopy that was not visible with other preoperative staging modalities.38 Current guidelines in Alberta recommend that staging laparoscopy be considered for all patients with potentially resectable GEA.39 A previous retrospective study by our group of 116 patients with GEA found that staging laparoscopy demonstrated M1 disease in 32% of cases initially deemed resectable on the basis of a CT scan.40 In the current study, staging laparoscopy revealed metastatic disease or positive cytology in 22% of the patients deemed to have resectable disease on staging CT, further supporting its usefulness in sparing patients noncurative surgery. Multivariable analysis in our population suggested that disease involving the entire stomach was associated with a significantly higher risk of a positive result on staging laparoscopy. We would therefore suggest that all patients with diffuse involvement of the entire stomach (linitis plastica) should undergo staging laparoscopy before any other treatment is initiated.
It has been clearly demonstrated that patients with intraperitoneal free cancer cells have a poorer prognosis.41 The prognosis for patients with suspicious cytology, as seen in 4 patients in our study, is less clear. Three of these patients went on to have curative-intent R0 resection but all 3 experienced recurrence and died at 463, 726 and 1083 days postoperatively, respectively. One patient did not have a resection as they were found to have metastatic disease on positron emission tomography (PET) scanning and died 107 days after diagnosis. The prognostic implication of suspicious cytology is a future area of interest for our group, and our transition to a national prospective clinical database will provide a larger sample size to study this issue.41
Current National Comprehensive Cancer Network (NCCN) guidelines support D2 lymph node resection if this can be performed with acceptable morbidity and mortality.3,42 In our study, 95% of procedures were completed by surgeons with appropriate fellowship-level training, and our median number of 22 lymph nodes harvested is higher than in recent Canadian studies.43,44 Sixty-two percent of our patients were found to have lymph node positive disease, similar to other North American studies.25,26 With regard to pathology, our rate of 82% R0 resection margins is similar to the 83% reported in a recent retrospective Canadian study by Levy and colleagues and comparable to the findings of earlier publications.26,45,46
Highly selected patients with GEA and peritoneal malignancy can experience long-term survival after cytoreductive surgery and HIPEC; however, mortality and morbidity rates remain high.47–49 Our study included 2 highly selected patients who had HIPEC, 1 of whom was alive with no known disease recurrence over 2 years after surgery and 1 of whom died 27 months after surgery. Both of these patients far outlived the median survival for patients with stage IV gastric cancer, reported as 8.9 months by the National Cancer Database (NCDB).50
In Canada, there is a relative paucity of recent data on survival outcomes for patients diagnosed with GEA. Available retrospective studies with data from the 1990s include a study from British Columbia, which reported an overall 5-year survival rate of 16% for all patients with GEA, and a study from our institution reporting 5-year disease specific survival rate of 28% in patients who received curative surgery.51,52 Ontario data from 2000–2005 suggested a 5-year survival rate for patients who had curative-intent surgery of 37%.18 More recent Canadian literature suggests a 3-year overall survival rate of 75% in patients who received curative treatment and achieved a “textbook outcome,” a metric used to indicate an optimal surgical course; patients who underwent resection without a textbook outcome had a 3-year overall survival rate of 55%.26 Considering differences in group stratification, these survival statistics parallel our reported 3-year overall survival rate of 63% for patients who received curative-intent surgery.
Limitations
Our study is limited by its relatively small sample size because of the rarity of GEA in Canada. Despite several attempts to get other centres in Alberta to enrol patients in this study, the vast majority of the patients were recruited from our academic centre at the University of Alberta. The sample size may explain why certain factors in our multivariable analysis such as age were not associated with increased mortality, which contradicts previous studies.53 In addition, given the voluntary prospective recruitment of patients, there was probably a selection bias toward patients from northern and central Alberta who were assessed by surgeons, and our sample may have been missing a population of patients who had been diagnosed with advanced, clearly unresectable disease. Our median follow-up time was only 14.5 months; we plan to continue to track our cohort of patients as the database matures, allowing further interpretation of survival. Finally, our study included 11 patients treated with distal esophagectomy who had carcinomas involving the GE junction. Given the similar histology and anatomic proximity, we feel it is reasonable to report these groups together. Recent studies suggest that many GEJ adenocarcinomas are molecularly similar to gastric adenocarcinomas and that more importance should be placed on considering histologic and molecular characteristics in making treatment decisions, rather than solely the location of the tumours.54,55
Conclusion
We have described a prospective gastroesophageal cancer database, which to our knowledge is the first of its kind in a Canadian setting. Our outcomes are similar to outcomes reported from other North American centres, demonstrating that patients with gastroesophageal cancer in Alberta present with advanced disease, with many being unable to receive curative-intent treatment. A high proportion of patients presenting with GEA at our centre had obesity, proximal tumours and signet ring histology. Those patients who were able to receive curative-intent surgery had comparable surgical and oncologic outcomes to similar populations across North America. In addition, our multivariable analysis supports the use of staging laparoscopy in all patients with diffuse malignancy involving the entire stomach. These findings will benefit Canadian patients directly and have led to academic collaboration across provinces and the development of a national prospective gastroesophageal cancer database involving multiple academic centres across Canada.
Acknowledgement:
The authors acknowledge Rose Cornand for her work in obtaining consent from patients and entering data for this study.
Footnotes
Presented at the 2022 International Gastric Cancer Congress, Mar. 6–9, 2022, Houston, Tex.
Competing interests: E. Bédard has received consulting fees from AstraZeneca and Hoffmann–La Roche. No other competing interests were declared.
Contributors: K. Stewart, M. McCall and D. Schiller designed the study. D. Skubleny, K. Stewart, S. Johnson and E. Haase acquired the data, which K. Purich, S. Ghosh and E. Bédard analyzed. K. Purich and E. Bédard wrote the article, which D. Skubleny, S. Ghosh, K. Stewart, S. Johnson, E. Haase, M. McCall and D. Schiller critically revised. All authors gave final approval of the version to be published.
- Accepted March 1, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/