Abstract
Background
We previously reported a high incidence of primary thyroid cancer in fluorodeoxyglucose positron-emission tomogram (FDG-PET)-detected incidental thyroid abnormalities. The aim of our study was to determine if these FDG-PET-detected thyroid malignancies represent a more-aggressive variant of primary thyroid carcinoma.
Materials and methods
All patients that underwent operative intervention for FDG-PET-detected incidental thyroid abnormalities were identified (June 2003 to April 2006). Patients with a diagnosis of primary thyroid carcinoma on final histopathology were included in the study. The patient demographics and histopathological findings were analyzed to identify adverse prognostic features.
Results
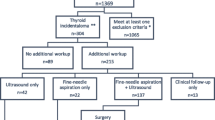
In 11,500 patients, 17,250 FDG-PET scans were performed; 377 of these patients (3.2% of patients and 2.1% of FDG-PET scans) had findings positive for thyroid abnormality. Of the 32 patients that underwent operative intervention, 22 patients with a final diagnosis of primary thyroid malignancy were included in the study. A greater number of patients [12 patients, (54%)] were noted to harbor poor prognostic variants of primary thyroid carcinoma on final histopathology [tall-cell variant: 11 patients (50%) and poorly differentiated thyroid carcinoma: 1 patient (4%)]. Extra-thyroidal extension (ETE) was noted in the majority of patients [14 patients (63%)]. In patients with tall cell variant on final histopathology, the rate of ETE was even higher [10 patients (90%)].
Conclusion
Thyroid malignancies incidentally detected on FDG-PET scan harbor a high rate of unfavorable prognostic features and may represent a more-aggressive variant of primary thyroid carcinoma. These patients need to be subjected to further investigation with a view to possible operative intervention.


Similar content being viewed by others
REFERENCES
Are C, Hsu JF, Schoder H, Shah JP, Larson SM, Shaha AR. FDG-PET detected thyroid incidentalomas: Need for further investigation? Ann Surg Oncol 2007;14(1):239–47
Cohen MS, Arslan N, Dehdashti F, Doherty G, Lairmore TC, Brunt M, Moley JF. Risk of malignancy in thyroid incidentalomas identified by fluorodeoxyglucose-positron emission tomography. Surgery 2001;130(6):941–946
Chen YK, Ding HJ, Chen KT, et al. Prevalence and risk of cancer of focal thyroid incidentalomas identified by 18F-Fluorodeoxyglusocse positron emission tomography for cancer screening in healthy subjects. Anticancer Res 2005;25:1421–1426
Kim TY, Kim WB, Ryu JS, Gong G, Hong SJ, Shong YK. 18F-Fluorodeoxyglusocse uptake in thyroid from positron emission tomogram (PET) for evaluation in cancer patients: high prevalence of malignancy in thyroid PET incidentalomas. Laryngoscope 2005;115:1074–1078
Ishimori T, Patel PV, Wahl RL. Detection of unexpected additional primary malignancies with FDG-PET/CT. J Nucl Med 2005;46(5):752–757
Kang KW, Kim SK, Kang HS, et al. Prevalence and risk of cancer of focal thyroid incidentalomas identified by 18F-Fluorodeoxyglusocse positron emission tomography for metastasis evaluation and cancer screening in healthy subjects. J Clin Endo Met 2005;88(9):4100–4104
Chu QD, Connor MS, Lilien DL, Johnson LW, Turnage RH, Li BD. Positron emission tomography (PET) positive thyroid incidentaloma: the risk of malignancy observed in a tertiary referral center. Am Surg 2006;72(3):272–5
Rosai J, Carcangiu, ML, DeLellis RA. Tumors of the thyroid gland. Atlas of tumor pathology. Washington, DC: Armed Forces Inst Pathol 1992:161–182
Bloom AD, Adler LP, Shuck JM. Determination of malignancy of thyroid nodules with positron emission tomography. Surgery 1993;114:728–735
Sasaki M, Ichiya Y, Kuwabara Y, et al. An evaluation of FDG-PET in the detection and differentiation of thyroid tumours. Nucl Med Commun 1997;18:957–963
Umetasu J, Sadato N, Ohtsubo T, et al. Fluorine-18 – fluorodeoxyglucose PET versus thallium-201 scintigraphy evaluation of thyroid tumours. J Nucl Med 1998;39:453–459
Gasparoni P, Rubello D, Ferlin G. Potential role of fluorine-18- fluorodeoxyglucose (FDG) positron emission tomography (PET) in the staging of primitive and recurrent medullary thyroid carcinoma. J Endocrinol Invest 1997;20:527–530
Feine U, Leitznmayer R, hanke J-P, et al. Flourine -18-FDG and iodine-131 iodide uptake in thyroid tissue. J Nucl Med 1996;37:1468–1472
de Geus-Oei LF, Pieters GF, Bonenkamp JJ, Mudde AH, Bleeker-Rovers CP, Corstens FH, Oyen WJ. 18 F-FDG PET reduces unnecessary hemithyroidectomies for thyroid nodules with inconclusive cytologic results. J Nucl Med 2006;47:770–775
Mitchell JC, Grant F, Evenson AR, Parker JA, Hasselgren PO, Parangi S. Pre operative evaluation of thyroid nodules with 18 FDG PET/CT. Surgery 2005;138:1166–1174
Are C, Shaha AR. Anaplastic thyroid carcinoma: Biology, Pathogenesis, Prognostic factors and treatment approaches. Ann Surg Oncol 2006;13:453–464
Lam AK, Lo CY, Lam KS. Papillary carcinoma of thyroid: A 30-yr clinicopathological review of the histological variants. Endocr Pathol Winter 2005;16(4):323–30
Machens A, Holzhausen HJ, Lautenschlager C, Dralle H. The tall-cell variant of papillary thyroid carcinoma: a multivariate analysis of clinical risk factors. Langenbecks Arch Surg 2004; 389(4):278–82
Michels JJ, Jacques M, Henry-Amar M, Bardet S. Prevalence and prognostic significance of tall cell variant of papillary thyroid carcinoma. Hum Pathol 2007;38(2):212–9
Johnson TL, Lloyd RV, Thompson NW, Beierwaltes WH, Sisson JC. Prognostic implications of the tall cell variant of papillary thyroid carcinoma. Am J Surg Pathol 1988;12(1):22–7
Prendiville S, Burman KD, Ringel MD, et al. Tall cell variant: an aggressive form of papillary thyroid carcinoma. Otolaryngol Head Neck Surg 2000;122(3):352–7
Siironen P, Louhimo J, Nordling S, Ristimaki A, Maenpaa H, Haapiainen R, Haglund C. Prognostic factors in papillary thyroid cancer: an evaluation of 601 consecutive patients. Tumour Biol 2005;26(2):57–64
Clark JR, Lai P, Hall F, Borglund A, Eski S, Freeman JL. Variables predicting distant metastases in thyroid cancer. Laryngoscope 2005;115(4):661–7
Bellantone R, Lombardi CP, Boscherini M, et al. Prognostic factors in differentiated thyroid carcinoma: a multivariate analysis of 234 consecutive patients. J Surg Oncol 1998;68(4):237–41
Andersen PE, Kinsella J, Loree TR, Shaha AR, Shah JP. Differentiated carcinoma of the thyroid with extrathyroidal extension. Am J Surg 1995;170(5):467–70
Shaha AR, Loree TR, Shah JP. Prognostic factors and risk group analysis in follicular carcinoma of the thyroid. Surgery 1995;118(6):1131–6; discussion 1136–8
Wreesmann VB, Sieczka EM, Socci ND. Genome-Wide Profiling of Papillary Thyroid Cancer Identifies MUC1 as an Independent Prognostic Marker. Cancer Res 2004;64:3780–3789
Sobrinho-Simoes MA, Nesland JM, Holm R, Sambade MC, Johannessen JV. Hurthle cell and mitochondrion-rich papillary carcinomas of the thyroid gland: an ultrastructural and immunocytochemical study. Ultrastruct Pathol 1985;8(2–3):131–42
S Mathupala P, Ko YH, Pedersen PL. Hexokinase II: Cancer’s double-edged sword acting as both facilitator and gatekeeper of malignancy when bound to mitochondria. Oncogene 2006;25:4777–4786
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Are, C., Hsu, J.F., Ghossein, R.A. et al. Histological Aggressiveness of Fluorodeoxyglucose Positron-Emission Tomogram (FDG-PET)-Detected Incidental Thyroid Carcinomas. Ann Surg Oncol 14, 3210–3215 (2007). https://doi.org/10.1245/s10434-007-9531-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-007-9531-4